 
The trigonal structure of ammonia is due to repulsive lone pair-bond pair attraction.The fourth full-filled hybridize orbital holds the lone pair of electrons.Three half-filled SP 3 orbitals of Nitrogen form a bond with three Hydrogen atoms.During hybridization, one s orbital and 3p orbitals hybridize to form four hybrid orbitals of equal energy levels, making SP 3 hybridization.Nitrogen in its ground state has configuration 1S 22S 22P 3.VEs = VEs in three Hydrogen atoms + VE in 1 Nitrogen atomĮlectronic Configuration of Nitrogen in Ammonia The table below shows the number of valence electrons in Ammonia Atom The NH3 molecule is held together by the strong N–H nitrogen–hydrogen single covalent bonds by sharing electrons. Three hydrogen atoms and one nitrogen atom combine to form ammonia so that the hydrogen atoms are electronically like helium and the nitrogen atom becomes like neon. NH3 Lewis structure shows that ammonia contains three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom.Įach hydrogen atom is missing one electron from a noble gas structure (complete shell), while nitrogen is missing three electrons from a full outer shell (of 8). 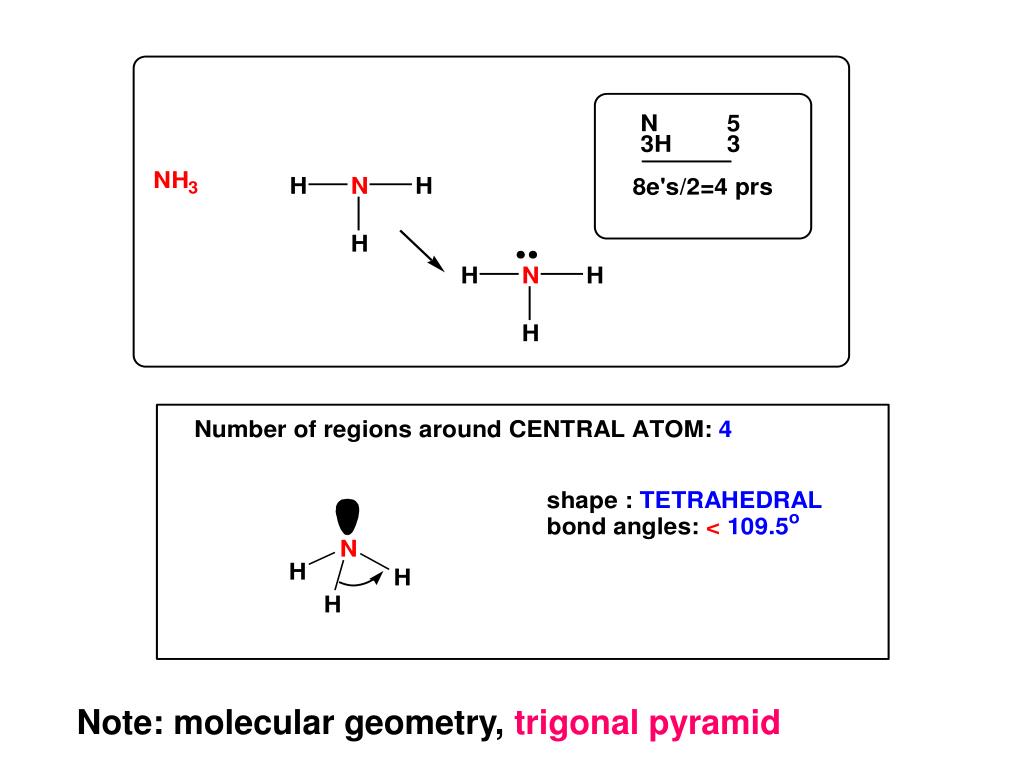
The unbonded electrons are called lone pairs of electrons. In NH3 molecular geometry, three hydrogen atoms are bonded to a nitrogen atom in the middle.īecause nitrogen has 5 electrons in its valence shell, it must interact with 3 hydrogen atoms to satisfy the octet rule and produce ammonia, a stable molecule. It is due to the presence of a single non-bonding lone pair of electrons on the nitrogen atom, which acts as a repulsive force on the bonding orbitals. What is the difference between ammonia and ammonium?Īmmonia (NH3) has a trigonal pyramidal or distorted tetrahedral molecular shape.The presence of monomeric Fe species in this Fe/ZSM-5 conveys molecular level insights into the temperature-dependent NH 3-SCR activity and might prove useful in the study of other reactions over monomeric Fe species, such as methane partial oxidation or dehydroaromatization reactions. Simulation of experimental X-ray absorption data revealed that the Fe species in the sample obtained by this approach adopt a square planar geometry, which changes into fivefold coordination at low temperature by strongly binding either NO or NH 3 as the ligand. Transient in situ XAS experiments at the Fe K-edge under dynamic NH 3-SCR reaction conditions demonstrated the involvement of all iron atoms in the redox cycle and, with that, suggest that Fe species in the studied Fe/ZSM-5 catalyst are monomers.  
Herein, we report on a preparation method to synthesize single-site Fe/ZSM-5 by combination of dealumination with the use of a bulky iron complex to introduce Fe. Monomeric Fe species in zeolites are considered to be the active sites for the low temperature activity toward ammonia-assisted selective catalytic reduction of nitrogen oxides (NH 3-SCR) in exhaust gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
